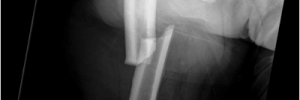
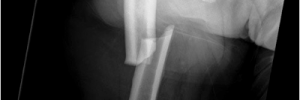
Alendronate is the most common medicine prescribed for the treatment of osteoporosis in the United States. Alendronate is a generic medicine made by various manufacturers in a tablet form. Now alendronate is available in an effervescent tablet called Binosto. Think Alka-Seltzer. Drop Binosto in room temperature water, it fizzes up and is in solution ready to drink after a quick stir.